Using cryo-electron microscopy, researchers at Case Western Reserve University School of Medicine and National Institutes of Health open door to new level of detail in study of abnormal proteins that cause untreatable neurodegenerative conditions
The highest-ever resolution imaging of an infectious prion has provided the first atomic-level data of how these abnormal proteins are assembled to cause fatal neurodegenerative diseases in people and animals—and how they can be potentially targeted by new therapies.
Conducted by Case Western Reserve University and the National Institutes of Health (NIH), the research is available through Molecular Cell. (The DOI for the paper: 10.1016/j.molcel.2021.08.011.)

“These detailed prion structures provide a new premise for understanding and targeting these currently untreatable diseases,” said Allison Kraus, lead and co-corresponding author of the research and an assistant professor in the Department of Pathology at Case Western Reserve University School of Medicine. “It will now be much easier to develop and test hypotheses about how prions are assembled as highly infectious and deadly protein structures.”
Seeing the basic building blocks of these lethal proteins, she said, provides a foundation for therapeutic strategies to block the spread, buildup and toxicity of prions.
Prions are proteins in brain tissue that transmit their irregular “misfolded” shapes onto the regular version of the same protein—and are the source of mammalian diseases, including human conditions like Creutzfeldt–Jakob disease (CJD) and its variant, known as vCJD, as well as Gerstmann–Sträussler–Scheinker syndrome, and others.
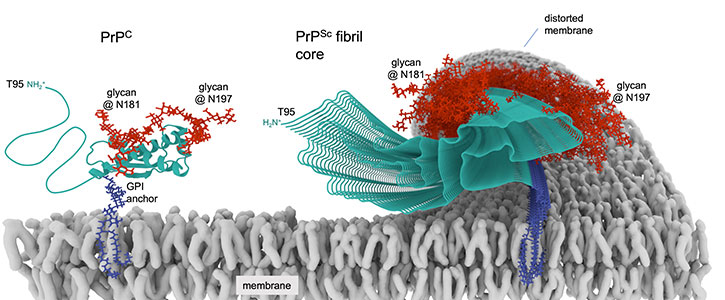
Similar prion-like mechanisms occur in the characteristic proteins suspected in the development of other neurodegenerative conditions, including Parkinson’s disease, Lou Gehrig’s disease (also known as ALS, or amyotrophic lateral sclerosis), chronic traumatic encephalopathy (CTE) and Alzheimer’s disease.
Though instances are rare, prion diseases can be transmitted between people; others are readily transmissible between animals, such as chronic wasting disease.
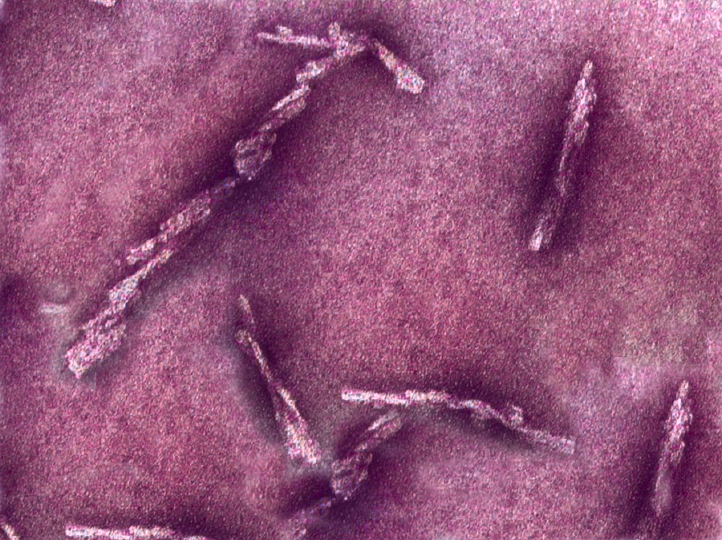
For this study, researchers imaged rodent-adapted scrapie prions derived from the brains of clinically ill hamsters.
New level of resolution
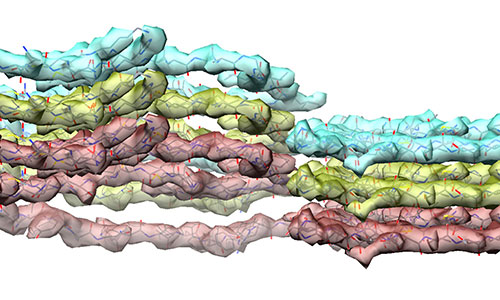
Using cryogenic-electron microscopy (cryo-EM)—at both NIH and the Cleveland Center for Structural and Membrane Biology Cryo-Electron Microscopy Core facilities at Case Western Reserve—and a collaborative pipeline between the Kraus (CWRU), Byron Caughey (NIH) and Research Technologies Branch (NIH) groups, researchers were able to determine aspects of the basic building blocks of these proteins, including the placements of their amino acids.
By suspending the prions in ice, cryo-electron technology allowed researchers to take thousands of images of the protein assemblies to build 3D atomic-resolution models using proprietary software.
This successful first-ever imaging to reach atomic-level detail of a brain-derived prion opens the door for similar “solving of other prion structures,” said Kraus. The study also obtained lower resolution images of another distinct prion strain that revealed structural differences between the two strains.
“It’s thought that there are many variations in prion structures as they relate to different diseases,” said Kraus. “Higher-resolution images provide clarity to many aspects of the cause and progression of these infectious diseases that are uniquely caused in nature by proteins—not viruses or bacteria.”
Co-authors of the research are: Forrest Hoyt, Cindi L. Schwartz, Bryan Hansen of NIH’s Rocky Mountain Laboratories (RML) Research Technologies Branch, and Efrosini Artikis, Andrew G. Hughson, Gregory J. Raymond, Brent Race, Gerald S. Baron and Caughey of RML’s Laboratory of Persistent Viral Diseases—both at the NIH’s National Institute of Allergy and Infectious Diseases in Hamilton, Montana.
For more information, contact Bill Lubinger at william.lubinger@case.edu.
This article was originally published Aug. 23, 2021.